The Gertrude’s Children’s Hospital news bulletin
Read Our News
2022 BO42452 – A Phase Ib, Randomized, Placebo-Controlled Study Evaluating The Safety, Pharmacokinetics, Pharmacodynamics, And Efficacy Of Crovalimab For The Management Of Acute Uncomplicated Vaso Occlusive Episodes (VOE) In Patients With Sickle Cell Disease (SCD)
2022 BO42452 – A Phase Ib, Randomized, Placebo-Controlled Study Evaluating The Safety, Pharmacokinetics, Pharmacodynamics, And Efficacy Of Crovalimab For The Management Of Acute Uncomplicated Vaso Occlusive Episodes (VOE) In Patients With Sickle Cell Disease (SCD)
2022 BO42451 – A Randomized Double-Blind Phase IIa Study Evaluating The Efficacy, Safety, Pharmacokinetics, And Pharmacodynamics Of Crovalimab As Adjunct Treatment In Prevention Of Vaso-Occlusive Episodes (Voe) In Sickle Cell Disease (SCD)
2022 BO42451 – A Randomized Double-Blind Phase IIa Study Evaluating The Efficacy, Safety, Pharmacokinetics, And Pharmacodynamics Of Crovalimab As Adjunct Treatment In Prevention Of Vaso-Occlusive Episodes (Voe) In Sickle Cell Disease (SCD)

Why Do People Get Kidney Stones in the Summer?
Why Do People Get Kidney Stones in the Summer?

6 Tips to Protect Your Mental Health When You’re Sick
6 Tips to Protect Your Mental Health When You’re Sick

Tips for Eating Healthy When You’re Working From Home
Tips for Eating Healthy When You’re Working From Home
New Parents
New Parents
5 Ways Your Child Can Stay Healthy
5 Ways Your Child Can Stay Healthy
As A Parent, Why Is Learning First Aid Important?
As A Parent, Why Is Learning First Aid Important?

Preparing For A Hospital Visit
Preparing For A Hospital Visit
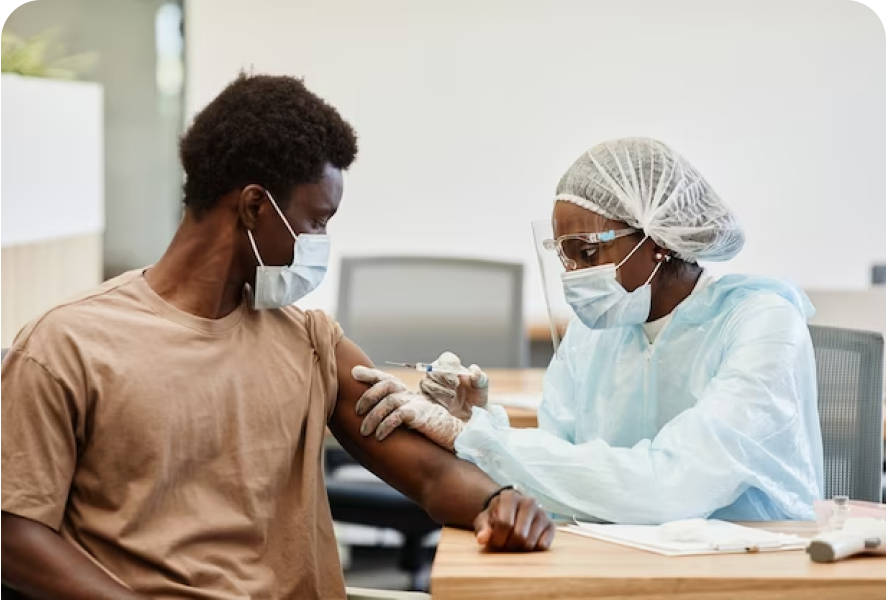
Community Health Screenings and Vaccination Clinics
Community Health Screenings and Vaccination Clinics

Support Groups and Counseling Services
Support Groups and Counseling Services

Health Education and Prevention Programs
Health Education and Prevention Programs

2012 TADO – A phase 3, Double-Blind, Randomized, Efficacy and Safety Comparison of Prasugrel and Placebo in Pediatric Patients with Sickle Cell Disease
2012 TADO – A phase 3, Double-Blind, Randomized, Efficacy and Safety Comparison of Prasugrel and Placebo in Pediatric Patients with Sickle Cell Disease

2015/2016 HESTIA 1 – Multicenter, Open-label, Randomized, Pharmacokinetic (PK) and Pharmacodynamic (PD) Dose-ranging, Phase 2 Study of Ticagrelor followed by a Double-blind, randomized, Parallel group, Placebo-controlled 4 weeks extension Phase in Pediatric Patients with Sickle Cell Disease
2015/2016 HESTIA 1 – Multicenter, Open-label, Randomized, Pharmacokinetic (PK) and Pharmacodynamic (PD) Dose-ranging, Phase 2 Study of Ticagrelor followed by a Double-blind, randomized, Parallel group, Placebo-controlled 4 weeks extension Phase in Pediatric Patients with Sickle Cell Disease

2018 HESTIA 4 – A Multi-centre, Phase 1, Open-label, Single-Dose Study to Investigate Pharmacokinetics (PK) of Ticagrelor in Infants and Toddlers, Aged 0 to less than 24 months, with Sickle Cell Disease
2018 HESTIA 4 – A Multi-centre, Phase 1, Open-label, Single-Dose Study to Investigate Pharmacokinetics (PK) of Ticagrelor in Infants and Toddlers, Aged 0 to less than 24 months, with Sickle Cell Disease
2019 HESTIA 3 – A Randomised, Double-Blind, Parallel-Group, Multi-centre, Phase 3 Study to Evaluate the Effect of Ticagrelor vs Placebo in Reducing the Rate of Vaso-Occlusive Crises in Pediatric Patients with Sickle Cell Disease
2019 HESTIA 3 – A Randomised, Double-Blind, Parallel-Group, Multi-centre, Phase 3 Study to Evaluate the Effect of Ticagrelor vs Placebo in Reducing the Rate of Vaso-Occlusive Crises in Pediatric Patients with Sickle Cell Disease
2017 GBT 031 – A Phase 3, Double-Blind, Randomized, Placebo-Controlled, Multi-Centre Study of GBT 440 Administered Orally to Patients with Sickle Cell Disease
2017 GBT 031 – A Phase 3, Double-Blind, Randomized, Placebo-Controlled, Multi-Centre Study of GBT 440 Administered Orally to Patients with Sickle Cell Disease
2018 GBT 034 – An Open-label Extension Study of Voxelotor (GBT 440) Administered Orally to Participants with Sickle Cell Disease Who Have Participated in Voxelotor Clinical Trials (GBT440-034)
2018 GBT 034 – An Open-label Extension Study of Voxelotor (GBT 440) Administered Orally to Participants with Sickle Cell Disease Who Have Participated in Voxelotor Clinical Trials (GBT440-034)
2020 IMR-SCD-301 ‘IMARA’ – A Phase 2b Study to Evaluate the Safety and Efficacy of IMR-687 in Subjects with Sickle Cell Disease
2020 IMR-SCD-301 ‘IMARA’ – A Phase 2b Study to Evaluate the Safety and Efficacy of IMR-687 in Subjects with Sickle Cell Disease
2021 GBT440-032 “HOPE Kids 2” – A Phase 3, Randomized, Double-Blind, Placebo Controlled Study of Voxelotor (GBT440) in Paediatric Participants with Sickle Cell Disease and an Open-label Study in Infants with Sickle Cell Disease
2021 GBT440-032 “HOPE Kids 2” – A Phase 3, Randomized, Double-Blind, Placebo Controlled Study of Voxelotor (GBT440) in Paediatric Participants with Sickle Cell Disease and an Open-label Study in Infants with Sickle Cell Disease
2022 GBT440-042 – Phase 3, Multicenter, Randomized, Double-Blind, PlaceboControlled Trial to Evaluate the Efficacy of Voxelotor for the Treatment of Leg Ulcers in Patients with Sickle Cell Disease
2022 GBT440-042 – Phase 3, Multicenter, Randomized, Double-Blind, PlaceboControlled Trial to Evaluate the Efficacy of Voxelotor for the Treatment of Leg Ulcers in Patients with Sickle Cell Disease

2021 GBT2104-131 – A Randomized, Double-blind, Placebo-controlled, Multicenter Study to Assess the Safety and Efficacy of Inclacumab in Participants with Sickle Cell Disease Experiencing Vasoocclusive Crises
2021 GBT2104-131 – A Randomized, Double-blind, Placebo-controlled, Multicenter Study to Assess the Safety and Efficacy of Inclacumab in Participants with Sickle Cell Disease Experiencing Vasoocclusive Crises

2022 GBT2104-133 – An Open-label Extension Study to Evaluate the Long-term Safety of Inclacumab Administered to Participants with Sickle Cell Disease Who Have Participated in an Inclacumab Clinical Trial
2022 GBT2104-133 – An Open-label Extension Study to Evaluate the Long-term Safety of Inclacumab Administered to Participants with Sickle Cell Disease Who Have Participated in an Inclacumab Clinical Trial
2021 AG348-C-020 ‘Agios’ – Phase 2/3, Double-Blind, Randomized, Placebo-Controlled, Multicenter Study to Evaluate the Efficacy and Safety of Mitapivat in Subjects With Sickle Cell Disease
2021 AG348-C-020 ‘Agios’ – Phase 2/3, Double-Blind, Randomized, Placebo-Controlled, Multicenter Study to Evaluate the Efficacy and Safety of Mitapivat in Subjects With Sickle Cell Disease
2021 FORMA-4202-HEM-301 – An Adaptive, Randomized, Placebo-controlled, Double-blind, Multi-center Study of Oral FT-4202, a Pyruvate Kinase Activator in Patients with Sickle Cell Disease
2021 FORMA-4202-HEM-301 – An Adaptive, Randomized, Placebo-controlled, Double-blind, Multi-center Study of Oral FT-4202, a Pyruvate Kinase Activator in Patients with Sickle Cell Disease
View our Media
The Gertrude’s Children’s Hospital media centre

HPV Vaccine Booking
HPV Vaccine Booking
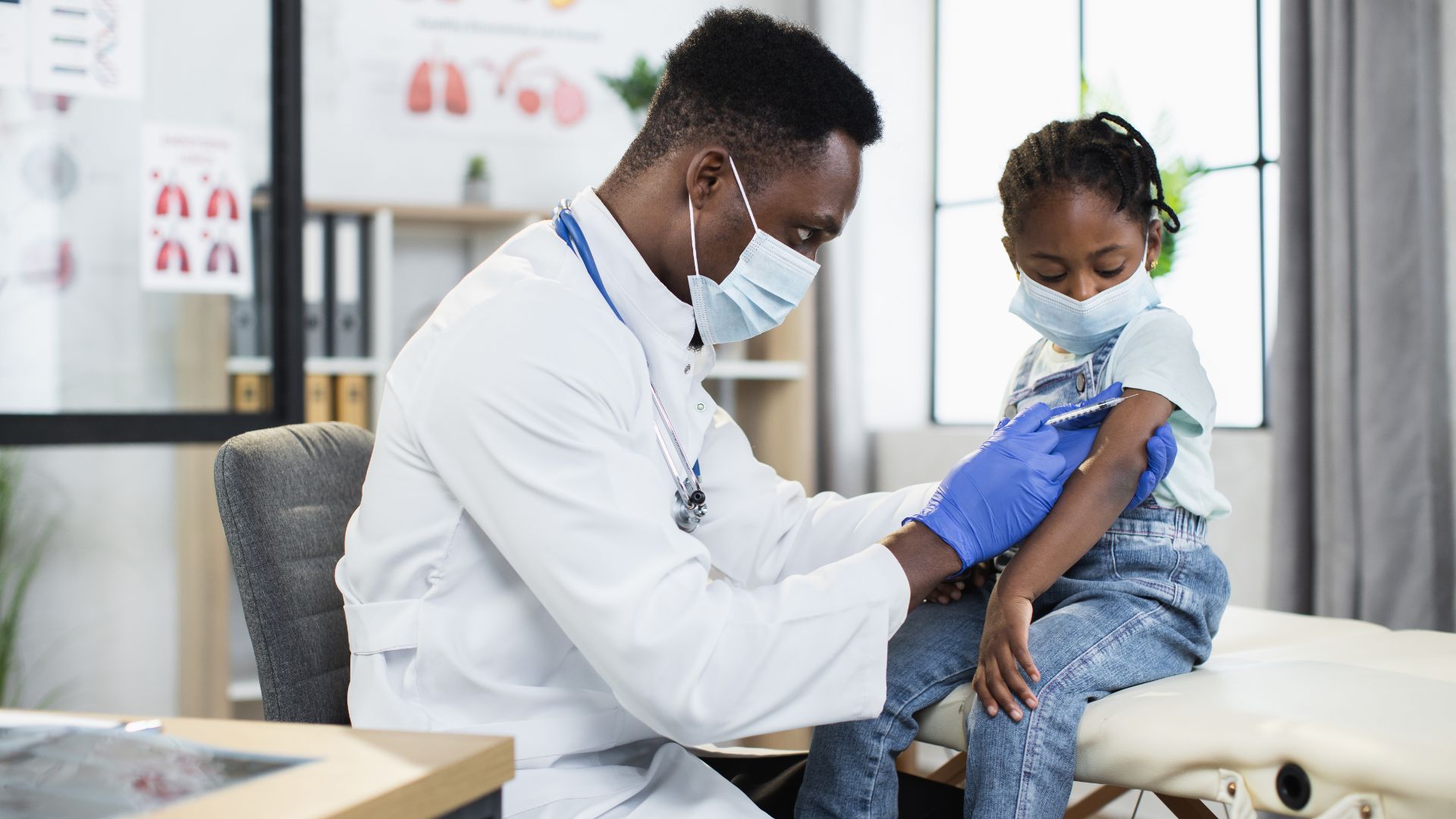
COVID-19 Vaccination Technical Guidance
COVID-19 Vaccination Technical Guidance

Pediatric Health Education Day for Teens
Pediatric Health Education Day for Teens

Family Support Workshop Series
Family Support Workshop Series

Pediatric Health and Wellness Fair
Pediatric Health and Wellness Fair
Our Downloads
The Gertrude’s Children’s Hospital downloads section
Githang’a, J. N., and P. Dave. “Bone marrow examination at a paediatric hospital in Kenya.” East African medical journal 78, no. 7 (2001): 37-39.
Githang’a, J. N., and P. Dave. “Bone marrow examination at a paediatric hospital in Kenya.” East African medical journal 78, no. 7 (2001): 37-39.
Forbes C, Hawkes M, Nesbitt S. Stool viruses among paediatric patients from a Nairobi clinic, Kenya. East Afr Med J. 2004 Nov;81(11):562-7. PMID: 15868964.
Forbes C, Hawkes M, Nesbitt S. Stool viruses among paediatric patients from a Nairobi clinic, Kenya. East Afr Med J. 2004 Nov;81(11):562-7. PMID: 15868964.
Onyango JF, Noah S. Pattern of clefts of the lip and palate managed over a three year period at a Nairobi hospital in Kenya. East Afr Med J. 2005 Dec;82(12):649-51. doi: 10.4314/eamj.v82i12.9371. PMID: 16619710.
Onyango JF, Noah S. Pattern of clefts of the lip and palate managed over a three year period at a Nairobi hospital in Kenya. East Afr Med J. 2005 Dec;82(12):649-51. doi: 10.4314/eamj.v82i12.9371. PMID: 16619710.
Wambani, J. S., G. K. Korir, I. K. Korir, and S. Kilaha. “Establishment of local diagnostic reference levels in paediatric screen-film radiography at a children’s hospital.” Radiation protection dosimetry 154, no. 4 (2013): 465-476.
Wambani, J. S., G. K. Korir, I. K. Korir, and S. Kilaha. “Establishment of local diagnostic reference levels in paediatric screen-film radiography at a children’s hospital.” Radiation protection dosimetry 154, no. 4 (2013): 465-476.
Makworo DG, Bwibo N, Omoni G. PARENTS’ AND HEALTHCARE PROVIDERS PERSPECTIVES ABOUT HOSPITALISED CHILDREN BEING VISITED BY OTHER CHILDREN IN NAIROBI, KENYA. East Afr Med J. 2013 Apr;90(4):108-16. PMID: 26866094.
Makworo DG, Bwibo N, Omoni G. PARENTS’ AND HEALTHCARE PROVIDERS PERSPECTIVES ABOUT HOSPITALISED CHILDREN BEING VISITED BY OTHER CHILDREN IN NAIROBI, KENYA. East Afr Med J. 2013 Apr;90(4):108-16. PMID: 26866094.
Omuse, Geoffrey, Beatrice Kabera, and Gunturu Revathi. “Low prevalence of methicillin resistant Staphylococcus aureus as determined by an automated identification system in two private hospitals in Nairobi, Kenya: a cross sectional study.” BMC infectious diseases 14 (2014): 1-6.
Omuse, Geoffrey, Beatrice Kabera, and Gunturu Revathi. “Low prevalence of methicillin resistant Staphylococcus aureus as determined by an automated identification system in two private hospitals in Nairobi, Kenya: a cross sectional study.” BMC infectious diseases 14 (2014): 1-6.
Welday, Seble Haile, Andrew Nyerere Kimang’a, Beatrice Muthoni Kabera, Josphine Wahogo Mburu, Christina Mwachari, Elizabeth Mungai, Stanley Mugambi Ndwiga, Joseph Kariuki Mbuthia, and Gunturu Revathi. “Stool as appropriate sample for the diagnosis of Mycobacterium tuberculosis by Gene Xpert test.” Open Journal of Respiratory Diseases 2014 (2014).
Welday, Seble Haile, Andrew Nyerere Kimang’a, Beatrice Muthoni Kabera, Josphine Wahogo Mburu, Christina Mwachari, Elizabeth Mungai, Stanley Mugambi Ndwiga, Joseph Kariuki Mbuthia, and Gunturu Revathi. “Stool as appropriate sample for the diagnosis of Mycobacterium tuberculosis by Gene Xpert test.” Open Journal of Respiratory Diseases 2014 (2014).
Mwangi, Beatrice. “Prevalence of chronic rhinosinusitis in children with dyspepsia at Kenyatta national hospital and Gertrude children’s hospital.” PhD diss., University of Nairobi, 2014.
Mwangi, Beatrice. “Prevalence of chronic rhinosinusitis in children with dyspepsia at Kenyatta national hospital and Gertrude children’s hospital.” PhD diss., University of Nairobi, 2014.
Mutiso VM, Muoki AS, Kimeu MM. PATTERNS OF POISONING AMONG PATIENTS AGED 0-13 YEARS AT A PAEDIATRIC HOSPITAL IN NAIROBI. East Afr Med J. 2014 Nov;91(11):379-84. PMID: 26866085.
Mutiso VM, Muoki AS, Kimeu MM. PATTERNS OF POISONING AMONG PATIENTS AGED 0-13 YEARS AT A PAEDIATRIC HOSPITAL IN NAIROBI. East Afr Med J. 2014 Nov;91(11):379-84. PMID: 26866085.
Rashid, Zahra Aly, Michel Millodot, and Katharine SE Evans. “Characteristics of keratoconic patients attending a specialist contact lens clinic in Kenya.” Middle East African journal of ophthalmology 23, no. 4 (2016): 283-287.
Rashid, Zahra Aly, Michel Millodot, and Katharine SE Evans. “Characteristics of keratoconic patients attending a specialist contact lens clinic in Kenya.” Middle East African journal of ophthalmology 23, no. 4 (2016): 283-287.
Odundo, Gordon Otieno. “428 Advocacy for enhanced scholar transport policies to prevent child injury: a case study from Kenya.” (2016): A156-A156.
Odundo, Gordon Otieno. “428 Advocacy for enhanced scholar transport policies to prevent child injury: a case study from Kenya.” (2016): A156-A156.
Odundo, Gordon Otieno, Thomas Ngwiri, Olivia Otuoma, Paul Laigong, Renson Mukhwana, Mary Slessor Limbe, and Nadia Musimbi Chanzu. “The impact and successes of a paediatric endocrinology fellowship program in Africa.” International journal of endocrinology 2016, no. 1 (2016): 1560248.
Odundo, Gordon Otieno, Thomas Ngwiri, Olivia Otuoma, Paul Laigong, Renson Mukhwana, Mary Slessor Limbe, and Nadia Musimbi Chanzu. “The impact and successes of a paediatric endocrinology fellowship program in Africa.” International journal of endocrinology 2016, no. 1 (2016): 1560248.
Migowa, Angela, Inés Colmegna, Carol Hitchon, Eugene Were, Evelyn Ng’ang’a, Thomas Ngwiri, John Wachira, Sasha Bernatsky, and Rosie Scuccimarri. “The spectrum of rheumatic in-patient diagnoses at a pediatric hospital in Kenya.” Pediatric Rheumatology 15 (2017): 1-6.
Migowa, Angela, Inés Colmegna, Carol Hitchon, Eugene Were, Evelyn Ng’ang’a, Thomas Ngwiri, John Wachira, Sasha Bernatsky, and Rosie Scuccimarri. “The spectrum of rheumatic in-patient diagnoses at a pediatric hospital in Kenya.” Pediatric Rheumatology 15 (2017): 1-6.
Agutu, Mary-Theresa, Julliette Ongus, Janeth Kombich, Rose Kamenwa, James Nyangao, John Kagira, Adelaide Ayoyi Ogutu, and Austine Bitek. “Prevalence and genetic diversity of rotavirus infection in children with acute gastroenteritis in a hospital setting, Nairobi Kenya in post vaccination era: a cross-sectional study.” Pan African Medical Journal 26, no. 1 (2017): 1-12.
Agutu, Mary-Theresa, Julliette Ongus, Janeth Kombich, Rose Kamenwa, James Nyangao, John Kagira, Adelaide Ayoyi Ogutu, and Austine Bitek. “Prevalence and genetic diversity of rotavirus infection in children with acute gastroenteritis in a hospital setting, Nairobi Kenya in post vaccination era: a cross-sectional study.” Pan African Medical Journal 26, no. 1 (2017): 1-12.
Hsu, Lewis L., Sharada Sarnaik, Suzan Williams, Carl Amilon, Jenny Wissmar, Anders Berggren, and HESTIA1 Investigators. “A dose‐ranging study of ticagrelor in children aged 3‐17 years with sickle cell disease: A 2‐part phase 2 study.” American Journal of Hematology 93, no. 12 (2018): 1493-1500.
Hsu, Lewis L., Sharada Sarnaik, Suzan Williams, Carl Amilon, Jenny Wissmar, Anders Berggren, and HESTIA1 Investigators. “A dose‐ranging study of ticagrelor in children aged 3‐17 years with sickle cell disease: A 2‐part phase 2 study.” American Journal of Hematology 93, no. 12 (2018): 1493-1500.
Kimani‐Murage, Elizabeth Wambui, Milka Njeri Wanjohi, Eva Watiri Kamande, Teresia Njoki Macharia, Elizabeth Mwaniki, Taddese Zerfu, Abdhalah Ziraba et al. “Perceptions on donated human milk and human milk banking in Nairobi, Kenya.” Maternal & Child Nutrition 15, no. 4 (2019): e12842.
Kimani‐Murage, Elizabeth Wambui, Milka Njeri Wanjohi, Eva Watiri Kamande, Teresia Njoki Macharia, Elizabeth Mwaniki, Taddese Zerfu, Abdhalah Ziraba et al. “Perceptions on donated human milk and human milk banking in Nairobi, Kenya.” Maternal & Child Nutrition 15, no. 4 (2019): e12842.
Nyarangi, Wambugu Naomi. “Using lean operations to reduce wastes in the medication administration process at the Gertrude’s Children’s Hospital, Nairobi, Kenya.” PhD diss., Strathmore University, 2019.
Nyarangi, Wambugu Naomi. “Using lean operations to reduce wastes in the medication administration process at the Gertrude’s Children’s Hospital, Nairobi, Kenya.” PhD diss., Strathmore University, 2019.
Mbuthia, J. “Comparison of hepatitis B immunity in HIV infected and uninfected kenyan children following primary immunization against hepatitis B virus.” International Journal of Infectious Diseases 101 (2020): 287.
Mbuthia, J. “Comparison of hepatitis B immunity in HIV infected and uninfected kenyan children following primary immunization against hepatitis B virus.” International Journal of Infectious Diseases 101 (2020): 287.
Mutua, Priscilla M. “Customer Service Strategy and competitive advantage of Getrudes’ Children’s Hospital.” PhD diss., University of Nairobi, 2020.
Mutua, Priscilla M. “Customer Service Strategy and competitive advantage of Getrudes’ Children’s Hospital.” PhD diss., University of Nairobi, 2020.
Ombuya, P., A. Barasa, and A. Laving. “Patterns of sensitization to food and inhalant allergens amongst children in Kenya.” East African Medical Journal 98, no. 7 (2021).
Ombuya, P., A. Barasa, and A. Laving. “Patterns of sensitization to food and inhalant allergens amongst children in Kenya.” East African Medical Journal 98, no. 7 (2021).
Omondi, Grace Petrine Oyito. “Clinicians’ perceptions of pediatric assent for invasive Bedside procedures at three tertiary referral Hospitals in Nairobi county, Kenya.” (2021).
Omondi, Grace Petrine Oyito. “Clinicians’ perceptions of pediatric assent for invasive Bedside procedures at three tertiary referral Hospitals in Nairobi county, Kenya.” (2021).
Patil RK, Irungu A, Kabera B, Mutua DK, Manguyu W, Lagat DK, Mutua KC. Severe combined immunodeficiency: a case series from a paediatric hospital in Kenya. Pan Afr Med J. 2021 May 20;39:56. doi: 10.11604/pamj.2021.39.56.26419. PMID: 34422179; PMCID: PMC8363958.
Patil RK, Irungu A, Kabera B, Mutua DK, Manguyu W, Lagat DK, Mutua KC. Severe combined immunodeficiency: a case series from a paediatric hospital in Kenya. Pan Afr Med J. 2021 May 20;39:56. doi: 10.11604/pamj.2021.39.56.26419. PMID: 34422179; PMCID: PMC8363958.
Patil RK, Kabera B, Muia CK, Ale BM. Hospital acquired infections in a private paediatric hospital in Kenya: a retrospective cross-sectional study. Pan Afr Med J. 2022 Jan 11;41:28. doi: 10.11604/pamj.2022.41.28.25820. PMID: 35291367; PMCID: PMC8895558.
Patil RK, Kabera B, Muia CK, Ale BM. Hospital acquired infections in a private paediatric hospital in Kenya: a retrospective cross-sectional study. Pan Afr Med J. 2022 Jan 11;41:28. doi: 10.11604/pamj.2022.41.28.25820. PMID: 35291367; PMCID: PMC8895558.
Zeinab, Ahmed. “Willingness and Perception of Caregivers on Covid-19 Vaccination of Their Children Under 12 Years in Tertiary Hospitals in Kenya.” PhD diss., University of Nairobi, 2023.
Zeinab, Ahmed. “Willingness and Perception of Caregivers on Covid-19 Vaccination of Their Children Under 12 Years in Tertiary Hospitals in Kenya.” PhD diss., University of Nairobi, 2023.
Juma, Bella. “Factors Affecting Haematological Parameters Before and After Introduction of Dolutegravir in Hiv-infected Children at Getrudes Childrens Hospital Comprehensive Care Clinic.” PhD diss., University of Nairobi, 2023.
Juma, Bella. “Factors Affecting Haematological Parameters Before and After Introduction of Dolutegravir in Hiv-infected Children at Getrudes Childrens Hospital Comprehensive Care Clinic.” PhD diss., University of Nairobi, 2023.

Waithaka, Rachael Wangeci, Janet Kerubo Maranga, and Celestine Khalechi Makobe. “Characterization of Extended Spectrum Beta-lactamase and Carbapenamase Producing Enterobacteriaceae Causing Urinary Tract Infection among Children in Kenya: A Cross-Sectional Study.” Advances in Microbiology 14, no. 7 (2024): 351-365.
Waithaka, Rachael Wangeci, Janet Kerubo Maranga, and Celestine Khalechi Makobe. “Characterization of Extended Spectrum Beta-lactamase and Carbapenamase Producing Enterobacteriaceae Causing Urinary Tract Infection among Children in Kenya: A Cross-Sectional Study.” Advances in Microbiology 14, no. 7 (2024): 351-365.

Lihanda, Bevin L. “The Economic Outcome of Antimicrobial Stewardship in a Hospital Setting: the Case of Gertrude’s Childrens’ Hospital.” PhD diss., University of Nairobi, 2023.
Lihanda, Bevin L. “The Economic Outcome of Antimicrobial Stewardship in a Hospital Setting: the Case of Gertrude’s Childrens’ Hospital.” PhD diss., University of Nairobi, 2023.

Juma, Bella. “Factors Affecting Haematological Parameters Before and After Introduction of Dolutegravir in Hiv-infected Children at Getrudes Childrens Hospital Comprehensive Care Clinic.” PhD diss., University of Nairobi, 2023




